For adults with intermediate- or high-risk MF1
Once-daily dosing with JAKAFI XR™ (ruxolitinib)1
Dose optimization is key to maintaining the balance between safety and efficacy1
For adults with intermediate- or high-risk MF, the recommended starting doses of JAKAFI XR are based on platelet counts1
A CBC, including platelet count, must be performed before initiating JAKAFI XR
- Platelet count of 50 to <100 × 10⁹/L: 11 mg once daily
- Platelet count of 100 to 200 × 10⁹/L: 33 mg once daily
- Platelet count >200 × 10⁹/L: 44 mg once daily
Before initiating JAKAFI XR, inquire about past infections, including tuberculosis, herpes simplex, herpes zoster, and hepatitis B.
JAKAFI® (ruxolitinib) dosages
5 mg twice daily
10 mg twice daily
15 mg twice daily
20 mg twice daily
25 mg twice daily
JAKAFI XR dosages
11 mg once daily
22 mg once daily
33 mg once daily
44 mg once daily
55 mg once daily
Tablet images shown are for demonstration purposes only and do not represent actual medications.
Dose modifications should be considered based on monitoring complete blood count (CBC), including platelet counts, absolute neutrophil count (ANC), and hemoglobin; treatment interruption and restarting dosing; or other adverse reactions, as described in the Full Prescribing Information for JAKAFI and JAKAFI XR.
JAKAFI XR is also available in 22-mg and 55-mg tablets.
Taking JAKAFI XR
- JAKAFI XR is dosed orally and can be administered with or without food
- Patients must swallow JAKAFI XR tablets whole. Do not split, chew, or crush tablets
Special populations: Please refer to the Full Prescribing Information for starting dose and other dose modifications, and for when to avoid treatment in patients with renal or hepatic impairment and in those receiving concomitant strong CYP3A4 inhibitors or fluconazole.
Monitoring patients after initiation of JAKAFI XR is essential, especially during the first 12 weeks of therapy1
- A CBC must be performed every 2 to 4 weeks until doses are stabilized, and then as clinically indicated. Doses may be titrated based on safety and efficacy
- Assess lipid parameters approximately 8 to 12 weeks following initiation of JAKAFI XR
Missed dose
- If a dose is missed, patients should not take an additional dose but should take the next usual prescribed dose
Individualize dosing of JAKAFI XR to optimize the balance between safety and efficacy1
Managing anemia, thrombocytopenia, neutropenia, and bleeding
- Dose modifications, temporarily withholding JAKAFI XR, and/or transfusions may be required for patients developing anemia or thrombocytopenia
- Interrupt treatment for bleeding, neutropenia (ANC <0.5 × 109/L), or thrombocytopenia (based on starting platelet count)
MF=myelofibrosis.
The effectiveness and safety of JAKAFI XR have been established based on adequate and well-controlled studies of JAKAFI
JAKAFI XR received approval based on comprehensive data demonstrating bioequivalence to JAKAFI1
There are no contraindications for use of JAKAFI XR, including in patients with anemia1
In COMFORT-I, 46% of patients receiving JAKAFI had anemia at baseline; among these patients, mean Hb was 9.2 g/dL (range, 6.6 g/dL to 13.7 g/dL)2
Adverse events with JAKAFI XR are expected to be similar to JAKAFI.1
Dose may be increased in the case of an insufficient response1
Efficacy based on titrated dose
COMFORT-I: Mean Change in Spleen Volume by Dose at Week 243
Results were assessed with JAKAFI, not JAKAFI XR. Please see the Full Prescribing Information for JAKAFI XR dosing information.
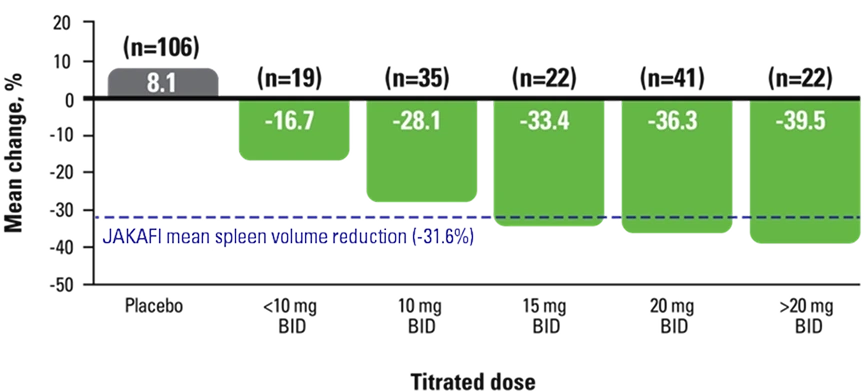
All patients in the clinical trials received JAKAFI twice daily.1
This material is under a CC BY-NC License and is the property of the Ferrata Storti Foundation. © 2026 Ferrata Storti Foundation. All rights reserved.
- Doses may be increased if the response is insufficient and platelet and neutrophil counts are adequate and treatment has not been reduced or interrupted in the prior 4 weeks1
- Doses should not be increased during the first 4 weeks of therapy and not more frequently than every 2 weeks1
- Discontinue JAKAFI XR if there is no spleen size reduction or symptom improvement after 6 months of therapy1
- Continuation of treatment for more than 6 months should be limited to patients in whom the benefits outweigh the potential risks1
BID=twice daily; COMFORT=COntrolled MyeloFibrosis study with ORal JAK inhibitor Treatment.
Dosing in COMFORT-I1
- Treatment with JAKAFI XR can cause thrombocytopenia, anemia, and neutropenia, which are each dose-related effects. Perform a pretreatment CBC and monitor CBCs every 2 to 4 weeks until doses are stabilized, and then as clinically indicated
- Manage thrombocytopenia by reducing the dose or temporarily interrupting JAKAFI XR. Platelet transfusions may be necessary
- Patients developing anemia may require blood transfusions and/or dose modifications of JAKAFI XR
- Severe neutropenia (ANC <0.5 × 109/L) was generally reversible by withholding JAKAFI XR until recovery
Anemia clinical trial data
Dose modifications of JAKAFI XR and/or blood transfusions may be required for patients developing anemia1
- In patients receiving JAKAFI in the COMFORT studies, mean decreases in Hb levels reached a nadir of approximately 1.5 g/dL to 2.0 g/dL below baseline after 8 to 12 weeks of therapy and then gradually recovered to a new steady state that was approximately 1.0 g/dL below baseline1
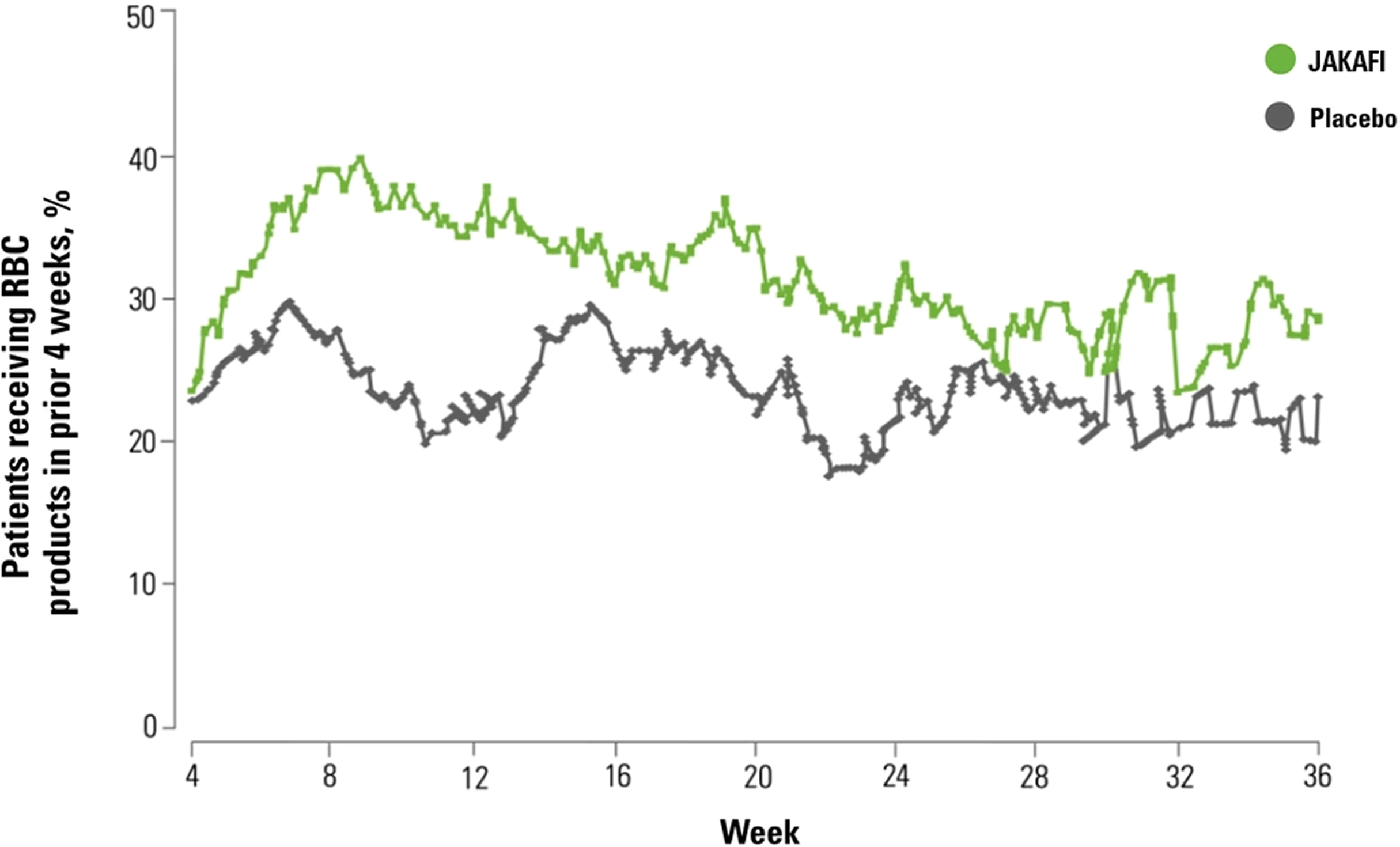
- In COMFORT-I, 60% of patients treated with JAKAFI and 38% of patients receiving placebo had RBC transfusions during randomized treatment1
COMFORT-I: Mean Hb Levels Over Time4
Results were assessed with JAKAFI, not JAKAFI XR. Please see the Full Prescribing Information for JAKAFI XR dosing information.
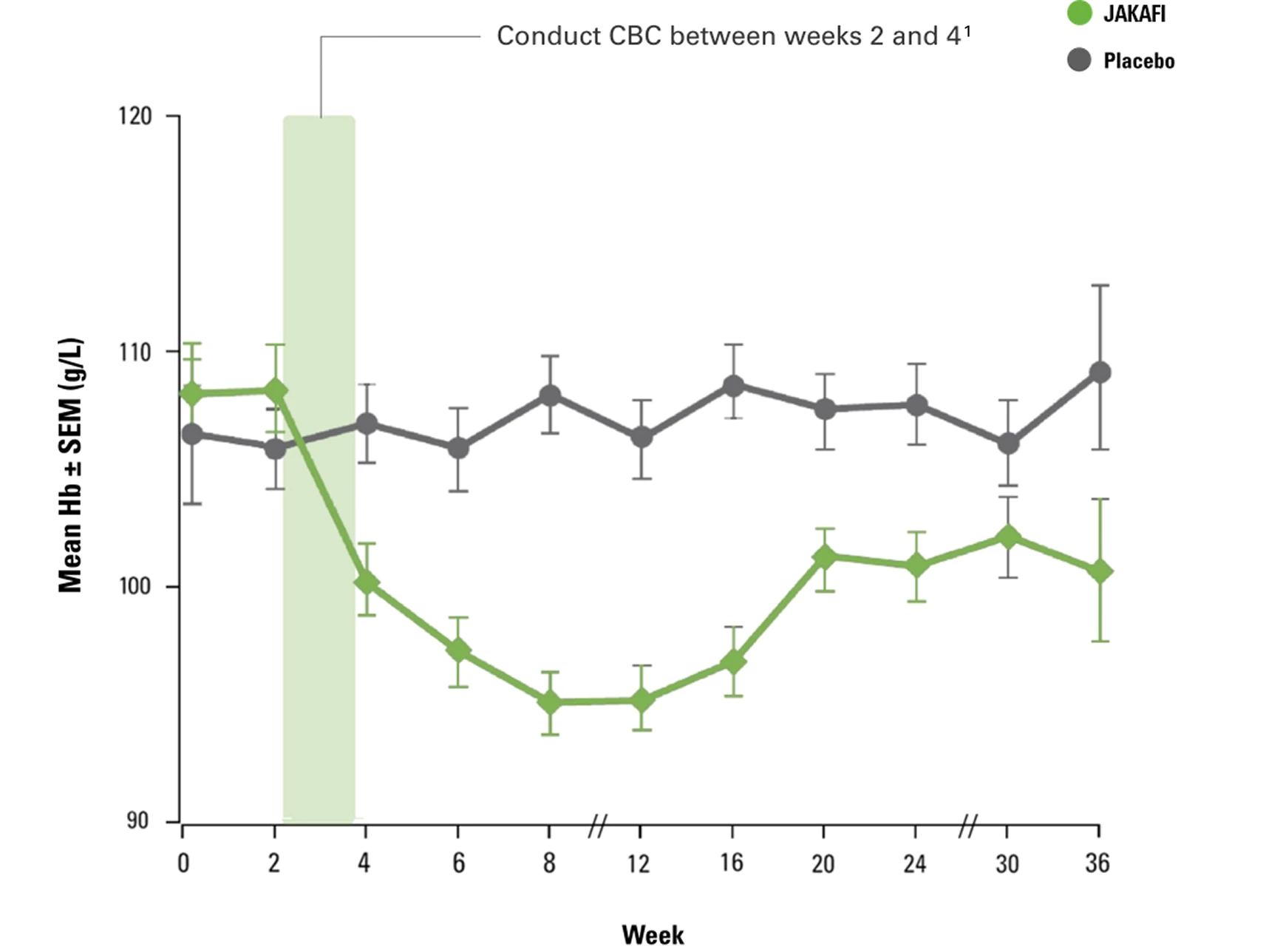
From The New England Journal of Medicine, Verstovsek S, Mesa RA, Gotlib J, et al, A double-blind, placebo-controlled trial of ruxolitinib for myelofibrosis, 366(9):799-807. Supplementary appendix available at: doi:10.1056/NEJMoa1110557. © 2012 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
- In COMFORT-I, grade 3 or 4 anemia occurred in 45% of patients receiving JAKAFI1
- <1% of patients receiving JAKAFI in the COMFORT studies discontinued due to anemia or thrombocytopenia4
COMFORT-I: Patients Requiring RBC Transfusions3
Results were assessed with JAKAFI, not JAKAFI XR. Please see the Full Prescribing Information for JAKAFI XR dosing information.
This material is under a CC BY-NC License and is the property of the Ferrata Storti Foundation. © 2026 Ferrata Storti Foundation. All rights reserved.
All patients in the clinical trials received JAKAFI twice daily. Adverse events with JAKAFI XR once daily are expected to be similar to JAKAFI twice daily.1
ANC=absolute neutrophil count; CBC=complete blood count; COMFORT=COntrolled MyeloFibrosis study with ORal JAK inhibitor Treatment; Hb=hemoglobin; RBC=red blood cell; SEM=standard error of the mean.
Thrombocytopenia clinical trial data
In patients with cytopenias, consider dose reductions, temporarily withholding JAKAFI XR, or transfusions as clinically indicated.1
Monitor CBCs during treatment, beginning as early as weeks 2 to 4.1
COMFORT-I: Mean Platelet Counts Over Time2
Results were assessed with JAKAFI, not JAKAFI XR. Please see the Full Prescribing Information for JAKAFI XR dosing information.
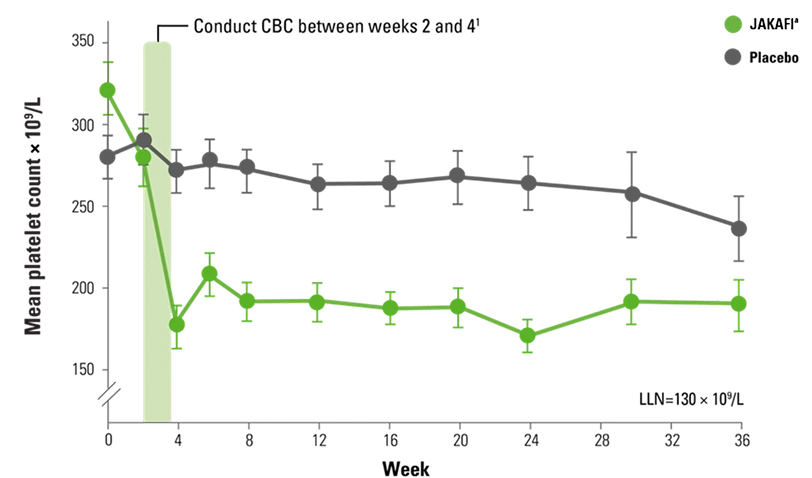
aProtocol-mandated dose modifications occurred based on platelet count.
- In COMFORT-I, grade 3 or 4 thrombocytopenia occurred in 13% of patients receiving JAKAFI1
- Initial reductions in Hb and platelets can occur in as early as 2 to 4 weeks2,4
- Dosing may need to be modified to avoid dose interruption, with the goal of achieving clinical benefit1
All patients in the clinical trials received JAKAFI twice daily. Adverse events with JAKAFI XR once daily are expected to be similar to JAKAFI twice daily.1
CBC=complete blood count; COMFORT=COntrolled MyeloFibrosis study with ORal JAK inhibitor Treatment; Hb=hemoglobin; LLN=lower limit of normal.
Appropriate dose management
In the case of a hematologic toxicity, including:
Thrombocytopenia
Discontinuations can be avoided by reducing the dose or temporarily withholding JAKAFI XR.
In patients receiving JAKAFI in the COMFORT studies, platelet counts and Hb levels generally stabilized after 8 to 12 weeks2,4
All patients in the clinical trials received JAKAFI twice daily. Adverse events with JAKAFI XR once daily are expected to be similar to JAKAFI twice daily.1
Anemia
Dose modifications of JAKAFI XR and/or blood transfusions may be required for patients developing anemia.1
COMFORT=COntrolled MyeloFibrosis study with ORal JAK inhibitor Treatment; Hb=hemoglobin.
Interrupt JAKAFI XR treatment for:
- Bleeding requiring intervention, regardless of current platelet count,
- Thrombocytopenia (platelet count <50 × 109/L), or
- Neutropenia (ANC <0.5 × 109/L)
Restarting after treatment interruption
- After recovery of platelet counts >50 × 109/L and ANC >0.75 × 109/L, dosing may be restarted
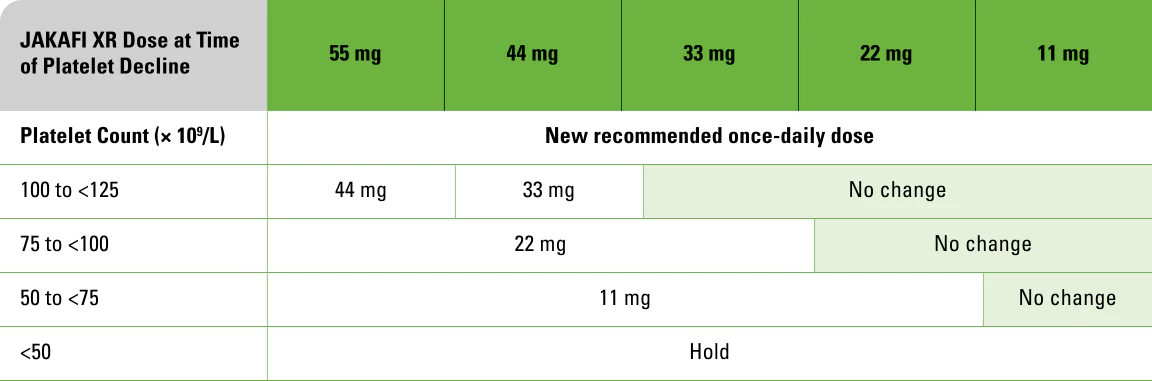
- Begin with a dose of JAKAFI XR that is at least 11 mg once daily below the dose at interruption
- The maximum allowable dose that may be used in restarting JAKAFI XR after a previous interruption is shown below
Maximum restarting doses for JAKAFI XR after safety interruption for thrombocytopenia

- Following treatment interruption for ANC <0.5 × 109/L, after ANC recovers to >0.75 × 109/L:
- For patients on 11 mg once daily prior to the first interruption, restart at 11 mg once daily. Discontinue for a second interruption
- For patients with a JAKAFI XR dose greater than 11 mg once daily prior to interruption, restart at 11 mg once daily below the largest dose in the week prior to interruption
ANC=absolute neutrophil count.
Increasing dose for insufficient response
In the case of an insufficient response, consider an increase in the dose if patient meets all of these criteria:
- Insufficient spleen reduction*
- Platelet count >125 × 109/L at 4 weeks and never <100 × 109/L
- ANC >0.75 × 109/L
*Failure to achieve a reduction from pretreatment baseline in either palpable spleen length of 50% or spleen volume of 35% as measured by CT or MRI.
Increase dose by increments of 11 mg once daily to a maximum of 55 mg once daily
Doses should not be increased during the first 4 weeks of therapy and should not be increased more frequently than every 2 weeks.
Discontinue JAKAFI XR if there is no spleen size reduction or symptom improvement after 6 months of therapy.
ANC=absolute neutrophil count; CT=computed tomography; MRI=magnetic resonance imaging.
Appropriate dose management
In the case of a hematologic toxicity, including:
Thrombocytopenia
Discontinuations can be avoided by reducing the dose or temporarily withholding JAKAFI XR.

In patients receiving JAKAFI in the COMFORT studies, platelet counts and Hb levels generally stabilized after 8 to 12 weeks2,4
All patients in the clinical trials received JAKAFI twice daily. Adverse events with JAKAFI XR once daily are expected to be similar to JAKAFI twice daily.1
Anemia
Dose modifications of JAKAFI XR and/or blood transfusions may be required for patients developing anemia.1
COMFORT=COntrolled MyeloFibrosis study with ORal JAK inhibitor Treatment; Hb=hemoglobin.
Interrupt JAKAFI XR treatment for:
- Bleeding requiring intervention, regardless of current platelet count,
- Thrombocytopenia (platelet count <50 × 109/L), or
- Neutropenia (ANC <0.5 × 109/L)
Restarting after treatment interruption
- After recovery of platelet counts >50 × 109/L and ANC >0.75 × 109/L, dosing may be restarted
- Begin with a dose of JAKAFI XR that is at least 11 mg once daily below the dose at interruption
- The maximum allowable dose that may be used in restarting JAKAFI XR after a previous interruption is shown below
Maximum restarting doses for JAKAFI XR after safety interruption for thrombocytopenia

- Following treatment interruption for ANC <0.5 × 109/L, after ANC recovers to >0.75 × 109/L:
- For patients on 11 mg once daily prior to the first interruption, restart at 11 mg once daily. Discontinue for a second interruption
- For patients with a JAKAFI XR dose greater than 11 mg once daily prior to interruption, restart at 11 mg once daily below the largest dose in the week prior to interruption
ANC=absolute neutrophil count.
Increasing dose for insufficient response
In the case of an insufficient response, consider an increase in the dose if patient meets all of these criteria:
- Insufficient spleen reduction*
- Platelet count >125 × 109/L at 4 weeks and never <100 × 109/L
- ANC >0.75 × 109/L
*Failure to achieve a reduction from pretreatment baseline in either palpable spleen length of 50% or spleen volume of 35% as measured by CT or MRI.
Increase dose by increments of 11 mg once daily to a maximum of 55 mg once daily
Doses should not be increased during the first 4 weeks of therapy and should not be increased more frequently than every 2 weeks.
Discontinue JAKAFI XR if there is no spleen size reduction or symptom improvement after 6 months of therapy.
ANC=absolute neutrophil count; CT=computed tomography; MRI=magnetic resonance imaging.
Decreasing dose for hematologic toxicity
Reduce the dose of JAKAFI XR in patients with platelet counts <35 × 109/L.

Interrupt JAKAFI XR treatment for:
- Bleeding requiring intervention, regardless of current platelet count,
- Thrombocytopenia (platelet count <25 × 109/L), or
- Neutropenia (ANC <0.5 × 109/L)
Restarting after treatment interruption
After treatment interruption, restart dosing after recovery of platelet counts to >35 × 109/L and ANC <0.75 × 109/L:
- Begin with a dose of JAKAFI XR that is at least 11 mg once daily below the largest dose in the week prior to the treatment interruption
- For patients on JAKAFI XR 11 mg once daily prior to the first interruption, continue JAKAFI XR 11 mg once daily
- Discontinue JAKAFI XR for a second interruption
ANC=absolute neutrophil count.
The recommended starting dose of JAKAFI XR in MF for patients with a starting platelet count of 50 to <100 × 109/L is 11 mg once daily
Increasing dose for insufficient response
In the case of an insufficient response, consider an increase in the dose only after the first 4 weeks of therapy and not more frequently than every 2 weeks if patients meet all of these criteria:
- Insufficient spleen reduction*
- Platelet count has remained ≥40 × 109/L at 4 weeks and has not decreased by >20% in the prior 4 weeks
- ANC >1.0 × 109/L
- No dose reduction or interruption for an AE or hematological toxicity in the prior 4 weeks
*Failure to achieve a reduction from pretreatment baseline in either palpable spleen length of 50% or spleen volume of 35% as measured by CT or MRI.
Increase dose by increments of 11 mg once daily to a maximum of 22 mg once daily
Continuation of treatment for more than 6 months should be limited to patients in whom the benefits outweigh the risks.
Discontinue JAKAFI XR if there is no spleen size reduction or symptom improvement after 6 months of therapy.
AE=adverse event; ANC=absolute neutrophil count; CT=computed tomography; MF=myelofibrosis; MRI=magnetic resonance imaging.
Decreasing dose for hematologic toxicity
Reduce the dose of JAKAFI XR in patients with platelet counts <35 × 109/L.

Interrupt JAKAFI XR treatment for:
- Bleeding requiring intervention, regardless of current platelet count,
- Thrombocytopenia (platelet count <25 × 109/L), or
- Neutropenia (ANC <0.5 × 109/L)
Restarting after treatment interruption
After treatment interruption, restart dosing after recovery of platelet counts to >35 × 109/L and ANC <0.75 × 109/L:
- Begin with a dose of JAKAFI XR that is at least 11 mg once daily below the largest dose in the week prior to the treatment interruption
- For patients on JAKAFI XR 11 mg once daily prior to the first interruption, continue JAKAFI XR 11 mg once daily
- Discontinue JAKAFI XR for a second interruption
ANC=absolute neutrophil count.
The recommended starting dose of JAKAFI XR in MF for patients with a starting platelet count of 50 to <100 × 109/L is 11 mg once daily
Increasing dose for insufficient response
In the case of an insufficient response, consider an increase in the dose only after the first 4 weeks of therapy and not more frequently than every 2 weeks if patients meet all of these criteria:
- Insufficient spleen reduction*
- Platelet count has remained ≥40 × 109/L at 4 weeks and has not decreased by >20% in the prior 4 weeks
- ANC >1.0 × 109/L
- No dose reduction or interruption for an AE or hematological toxicity in the prior 4 weeks
*Failure to achieve a reduction from pretreatment baseline in either palpable spleen length of 50% or spleen volume of 35% as measured by CT or MRI.
Increase dose by increments of 11 mg once daily to a maximum of 22 mg once daily
Continuation of treatment for more than 6 months should be limited to patients in whom the benefits outweigh the risks.
Discontinue JAKAFI XR if there is no spleen size reduction or symptom improvement after 6 months of therapy.
AE=adverse event; ANC=absolute neutrophil count; CT=computed tomography; MF=myelofibrosis; MRI=magnetic resonance imaging.
Dose modification for bleeding1
Interrupt treatment for bleeding requiring intervention regardless of current platelet count. Once the bleeding event has resolved, consider resuming treatment at the prior dose if the underlying cause of bleeding has been controlled. If the bleeding event has resolved but the underlying cause persists, consider resuming treatment with JAKAFI XR at a lower dose.
Dose modifications for special populations1
Dose modifications in patients with renal or hepatic impairment, or when coadministered with strong CYP3A4 inhibitors or fluconazole
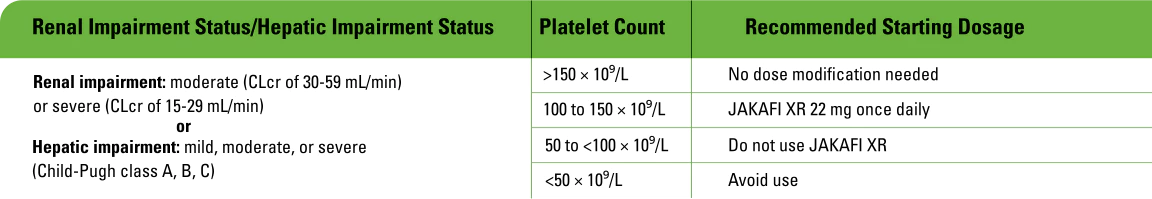
Patients with ESRD on dialysis
- Do not use JAKAFI XR for patients with MF who have ESRD (CLcr <15 mL/min) and are on dialysis with a platelet count between 100 × 109/L and 200 × 109/L or >200 × 109/L
- Avoid use of JAKAFI XR in patients with ESRD (CLcr <15 mL/min) not requiring dialysis
Concomitant use with strong CYP3A4 inhibitors or fluconazole
- Modify the dose of JAKAFI XR when coadministered with strong CYP3A4 inhibitors and fluconazole doses of ≤200 mg for MF
- Avoid the use of fluconazole doses >200 mg daily with JAKAFI XR
- Additional dose modifications should be made with frequent monitoring of safety and efficacy
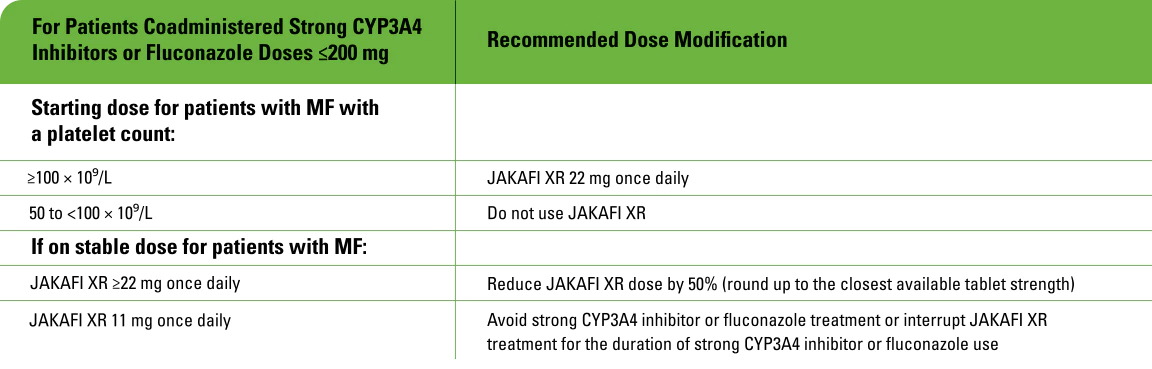
CLcr=creatinine clearance; ESRD=end-stage renal disease; MF=myelofibrosis.
COMFORT=COntrolled MyeloFibrosis study with ORal JAK inhibitor Treatment; Hb=hemoglobin; MF=myelofibrosis; MPN=myeloproliferative neoplasm; OS=overall survival; SVR=spleen volume reduction.
References: 1. JAKAFI/JAKAFI XR Prescribing Information. Wilmington, DE: Incyte Corporation. 2. Data on file. Incyte Corporation. Wilmington, DE. 3. Verstovsek S, Mesa RA, Gotlib J, et al. Efficacy, safety and survival with ruxolitinib in patients with myelofibrosis: results of a median 2-year follow-up of COMFORT-I. Haematologica. 2013;98(12):1865-1871. Supplementary appendix available at: https://haematologica.org/article/view/6861. 4. Verstovsek S, Mesa RA, Gotlib J, et al. A double-blind, placebo-controlled trial of ruxolitinib for myelofibrosis. N Engl J Med. 2012;366(9):799-807. Supplementary appendix available at: https://www.nejm.org/doi/full/10.1056/nejmoa1110557.