For PV in adults who have had an inadequate response to HU1
Individualized dosing for Jakafi® (ruxolitinib)1
Dose optimization is key to maintaining the balance between safety and efficacy
For patients with PV who have an inadequate response to HU, the recommended starting dose is 10 mg BID.
A CBC and platelet count must be performed before initiating Jakafi.

Jakafi is also available in tablets of:




A CBC and platelet count must be performed before initiating Jakafi, every 2 to 4 weeks until doses are stabilized, and then as clinically indicated.
Individualize dosing of Jakafi to optimize the balance between safety and efficacy.
- Dosing may be reduced or temporarily interrupted based on Hb, platelet, or neutrophil counts
- Additionally, dosing may be increased to achieve desired clinical response
- Interrupt treatment for bleeding
BID=twice daily; CBC=complete blood count; Hb=hemoglobin; HU=hydroxyurea; PV=polycythemia vera; RESPONSE=Randomized study of Efficacy and Safety in POlycythemia vera with JAK iNhibitor ruxolitinib verSus bEst available care.
Decrease dose for anemia or thrombocytopenia1

Among those taking Jakafi in the RESPONSE trial, 5% (6/110) of patients decreased from the starting dose at week 8, and 13% (14/110) of patients decreased from the starting dose at week 32.2
BID=twice daily; Hb=hemoglobin; RESPONSE=Randomized study of Efficacy and Safety in POlycythemia vera with JAK iNhibitor ruxolitinib verSus bEst available care.
Increase dose for insufficient response1
Doses may be increased in increments of 5 mg BID to a maximum of 25 mg BID
- Doses should not be increased during the first 4 weeks of therapy and should not be increased more frequently than every 2 weeks
Consider dose increases in patients who meet all of these criteria:
- Inadequate efficacy as demonstrated by 1 or more of the following:
- - Continued need for phlebotomy
- - WBC count greater than the ULN range
- - Platelet count greater than the ULN range
- - Palpable spleen that is reduced <25% from baseline
- Platelet count ≥140 × 109/L
- Hb ≥12 g/dL
- ANC ≥1.5 × 109/L
Among those taking Jakafi in the RESPONSE trial, 37% (41/110) of patients increased from the starting dose at week 8, and 54% (59/110) of patients increased from the starting dose at week 32.2
ANC=absolute neutrophil count; BID=twice daily; Hb=hemoglobin; RESPONSE=Randomized study of Efficacy and Safety in POlycythemia vera with JAK iNhibitor ruxolitinib verSus bEst available care; ULN=upper limit of normal; WBC=white blood cell.
Interrupting/restarting dosing1
Interrupt Jakafi treatment for:
- Anemia (Hb <8 g/dL),
- Thrombocytopenia (platelet count <50 × 109/L), or
- Neutropenia (ANC <1.0 × 109/L)
After recovery of hematologic parameter(s) to acceptable levels, dosing may be restarted
Use the most severe category of patients’ Hb, platelet count, or ANC abnormality to determine the corresponding maximum restarting dose:
- aContinue treatment for at least 2 weeks; if stable, may increase dose by 5 mg BID.
Patients who required dose interruption while receiving a dose of 5 mg BID may restart at a dose of 5 mg BID or 5 mg once daily, but not higher, when they have:
- Hb ≥10 g/dL,
- Platelet count ≥75 × 109/L, and
- ANC ≥1.5 × 109/L
Dose management after restarting treatment in PV
- After restarting Jakafi following treatment interruption, doses may be titrated, but the maximum total daily dose should not exceed 5 mg less than the dose that resulted in the dose interruption
- An exception to this is dose interruption following phlebotomy-associated anemia, in which case the maximal total daily dose allowed after restarting Jakafi would not be limited
Special populations:
- Avoid fluconazole doses >200 mg daily with Jakafi
- Avoid use of Jakafi in patients with end-stage renal disease (CLcr <15 mL/min) not requiring dialysis
ANC=absolute neutrophil count; BID=twice daily; CLcr=creatinine clearance; Hb=hemoglobin; PV=polycythemia vera.
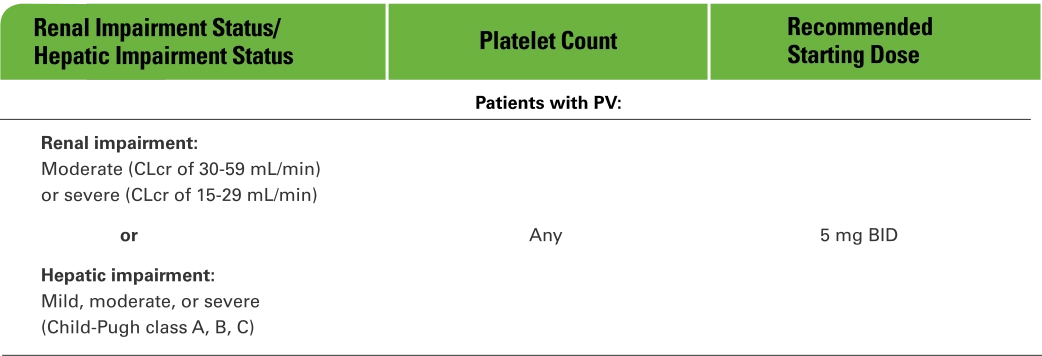
Dose modifications for special populations1
Modify the dose of Jakafi accordingly in patients with moderate or severe renal impairment, patients with hepatic impairment, and patients on dialysis.
Patients with end-stage renal disease on dialysis
- The recommended starting dose in patients with PV who have end-stage renal disease (CLcr <15 mL/min) and are on dialysis is 10 mg once after dialysis session
- Additional dose modifications should be made with frequent monitoring of safety and efficacy
- Avoid use of Jakafi in patients with end-stage renal disease (CLcr <15 mL/min) not requiring dialysis
Concomitant use with strong CYP3A4 inhibitors or fluconazole
- Modify the dose of Jakafi when coadministered with strong CYP3A4 inhibitors and fluconazole doses of ≤200 mg
- Avoid the use of fluconazole doses <200 mg daily with Jakafi
- Additional dose modifications should be made with frequent monitoring of safety and efficacy

BID=twice daily; CLcr=creatinine clearance; PV=polycythemia vera.
HU=hydroxyurea; MPN=myeloproliferative neoplasm; PV=polycythemia vera.
References: 1. Jakafi [package insert]. Wilmington, DE: Incyte Corporation. 2. Data on file. Incyte Corporation. Wilmington, DE.

