Perspectives and insights from PV experts
The resources on this page offer information and insights about Jakafi® (ruxolitinib) and treatment of patients with PV who have had an inadequate response to or are intolerant of HU. Be sure to check back regularly as new materials will be added as they become available.


MAJIC-PV study results: WBC control and risk of thrombosis
Dr Claire Harrison discusses findings from the RESPONSE and MAJIC-PV studies, including data on the correlation between WBC control and thrombosis-free survival.
Hello, my name is Claire Harrison, hematologist, and lead author of the MAJIC-PV study.
In addition to sharing information from the RESPONSE study, I’m excited to discuss several important findings from the MAJIC-PV study, published in the Journal of Clinical Oncology in 2023. These findings, I believe, may change the way we think about the treatment of polycythemia vera.
When it comes to managing my patients with PV, there are a variety of important factors that I keep in mind, as they can vary from patient to patient. In my practice, we communicate treatment goals in the context of longer-term risks, primarily thrombotic events.
As we continue to understand the underlying pathophysiology of atherosclerosis and thrombosis, we are learning that elevated white blood cell counts may have implications. While the correlation between the risk of thrombosis and elevated hematocrit has been well documented, controlling other parameters of blood count has been a bit controversial. An additional analysis from CYTO-PV found that there was a 4-fold increase in the risk of major thrombosis among patients with white blood cell counts above 11 compared to those with white blood cell counts below 7.
I find that it can be difficult to disentangle the different aspects of the blood count and I believe what these data are saying is that white blood cell counts at the top end of normal may convey an extra risk to the patient and that frequently these patients will be well above counts of 11, 15, even 20. So, if my patient is having 4 phlebotomies a year and their white blood cell counts are greater than 11 and progressing, that is concerning. That’s a disease with risk.
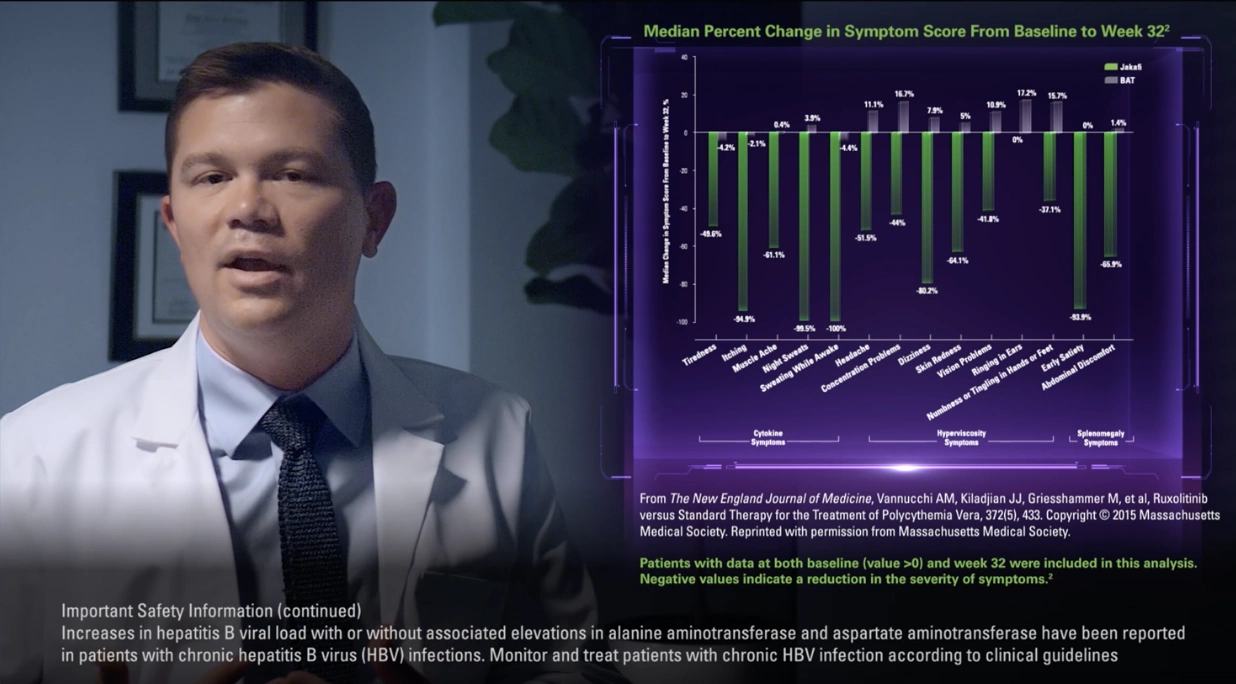
As previously demonstrated in the RESPONSE study, which evaluated hematocrit control and spleen volume reduction, 23% of patients who received ruxolitinib were able to achieve this combined primary endpoint compared to less than 1% of those who received best available therapy, the majority of whom received hydroxyurea. These results were achieved in the absence of phlebotomy.
But more than just achieving hematocrit control, the trial also included complete hematologic response as an endpoint. And it was found that 24% of patients receiving ruxolitinib achieved complete hematologic remission and those patients had a 55% probability of maintaining it at 5 years.
Here we have the RESPONSE safety profile, which includes nonhematologic adverse reactions occurring in more than or equal to 12% of patients and clinically relevant hematologic laboratory abnormalities.
In this study we also saw that any grade of thromboembolic events occurred in 0.9% of patients receiving ruxolitinib versus 5.4% with best available therapy.
For some time now, we have wanted to assess at a deeper level the data for ruxolitinib; however, the RESPONSE study involved crossover, and therefore certain endpoints could not be evaluated. This is why we conducted the MAJIC-PV study, which did not include crossover.
MAJIC-PV was an open-label, phase 2, randomized, controlled trial in which we compared ruxolitinib versus best available therapy in 180 patients with high-risk PV who met the criteria for being resistant or intolerant to hydroxyurea.
Patients were randomized to ruxolitinib or best available therapy, with hydroxyurea being the most frequently used treatment. In MAJIC-PV, there was no preplanned crossover to ruxolitinib, and patients were followed for 5 years. The primary endpoint was complete response rate within 12 months as defined by ELN criteria: hematocrit less than 45% without phlebotomy for 3 months; platelets less than or equal to 400; white blood cell count less than or equal to 10; and normal spleen size. Secondary exploratory endpoints included event-free survival and thrombosis-free survival. Although these endpoints were unpowered, we were able to assess them because there was no crossover in the study.
We knew from the CYTO-PV study that there was a correlation between increased white blood cell count and thrombotic risk, but before the MAJIC-PV study, we didn’t really understand the full relevance of controlling white cell count.
Our target with the MAJIC-PV study was to control blood counts, that is, achieve complete response, which included hematocrit of less than 45% without the need for phlebotomy, white blood cell count less than or equal to 10, and normal platelet count. In the study, 43% of patients receiving ruxolitinib achieved a complete response within 12 months versus 26% of patients receiving best available therapy, supporting what we already know about ruxolitinib from the RESPONSE study in terms of superior blood count control versus best available therapy.
We then extended the analysis to see if ruxolitinib had an impact on event-free survival, which was a composite of major thrombosis, major hemorrhage, transformation, or death. And what we found, for the first time in a prospective study, is that ruxolitinib demonstrated a 42% reduction in the risk of an event.
What stands out to me are the data on thrombosis-free survival. In the MAJIC-PV study, there was a 44% reduction in the risk of major thrombosis with ruxolitinib. As you can see, the separation between the curves happens early on and they remain separated throughout the study. It’s important to remember that inclusion criteria for the study was hydroxyurea resistance. So, from my perspective, these data tell me that my patients showing signs of hydroxyurea resistance may be at risk of thrombosis. Alongside these data, we presented the number of phlebotomies. Patients in the best-available-therapy arm underwent a greater number of phlebotomies versus those in the ruxolitinib arm: 307 and 83, respectively.
It’s important to remember that treatment goals were the same for both arms, to achieve a complete response without the need for phlebotomy. For the best-available-therapy arm, we instructed clinicians to treat their high-risk patients as they would treat them in their clinic. So, what these data also tell me is that we need to sit up and take notice, because these patients are at increased risk of major thrombosis. In my practice, for any patient on hydroxyurea who has hematocrit levels between 45% and 50%, has progressive leukocytosis, and/or receiving more than 4 phlebotomies a year, this is where I would consider changing their treatment regimen to an agent like ruxolitinib.
From a safety perspective, we didn’t see any new signals in the overall MAJIC-PV population. The safety profile was generally consistent with previous reports for ruxolitinib.
From the findings of these studies, I’d like for my colleagues to understand the importance of identifying patients who have hematocrit levels between 45% and 50%, white blood cell counts above 11, or are needing more than 4 phlebotomies a year. These are the warning signs that hydroxyurea is inadequate and we need to take action immediately and consider a change in treatment plan.
In my practice, I aim to control all aspects of the blood count: hematocrit less than 45% and white blood cell count less than 11. I find that when increasing the hydroxyurea dose, it may take weeks for levels to drop, and I wouldn’t necessarily wait until my patient was on 2 grams or 2.5 grams a day. For me, that’s a big dose. So, if I cannot increase the dose and they’ve needed 2 or more phlebotomies in the last 6 months, this is the point at which I begin to discuss a change in treatment. PV is quite a rare condition and oftentimes, colleagues have much sicker patients in their clinic, but the decisions we make for our patients with PV today may impact them for many years.
MAJIC-PV corroborated what we already knew about ruxolitinib in terms of controlling blood counts. You can see through the data that there is a benefit in having a strategy of controlling hematocrit and white blood cell count. It’s important for me, not only as the lead author of the MAJIC-PV study but also as a physician, to see these long-term data, and we now have the data to show reduction in the risk of thrombosis for these patients as well.
INDICATIONS AND USAGE
Jakafi® (ruxolitinib) is indicated for treatment of polycythemia vera (PV) in adults who have had an inadequate response to or are intolerant of hydroxyurea.
IMPORTANT SAFETY INFORMATION
- Treatment with Jakafi® (ruxolitinib) can cause thrombocytopenia, anemia and neutropenia, which are each dose-related effects. Perform a pre-treatment complete blood count (CBC) and monitor CBCs every 2 to 4 weeks until doses are stabilized, and then as clinically indicated
- Manage thrombocytopenia by reducing the dose or temporarily interrupting Jakafi. Platelet transfusions may be necessary
- Patients developing anemia may require blood transfusions and/or dose modifications of Jakafi
- Severe neutropenia (ANC <0.5 × 109/L) was generally reversible by withholding Jakafi until recovery
- Serious bacterial, mycobacterial, fungal and viral infections have occurred. Delay starting Jakafi until active serious infections have resolved. Observe patients receiving Jakafi for signs and symptoms of infection and manage promptly. Use active surveillance and prophylactic antibiotics according to clinical guidelines
- Tuberculosis (TB) infection has been reported. Observe patients taking Jakafi for signs and symptoms of active TB and manage promptly. Prior to initiating Jakafi, evaluate patients for TB risk factors and test those at higher risk for latent infection. Consult a physician with expertise in the treatment of TB before starting Jakafi in patients with evidence of active or latent TB. Continuation of Jakafi during treatment of active TB should be based on the overall risk-benefit determination
- Progressive multifocal leukoencephalopathy (PML) has occurred with Jakafi treatment. If PML is suspected, stop Jakafi and evaluate
- Herpes zoster infection has been reported in patients receiving Jakafi. Advise patients about early signs and symptoms of herpes zoster and to seek early treatment. Herpes simplex virus reactivation and/or dissemination has been reported in patients receiving Jakafi. Monitor patients for the development of herpes simplex infections. If a patient develops evidence of dissemination of herpes simplex, consider interrupting treatment with Jakafi; patients should be promptly treated and monitored according to clinical guidelines
- Increases in hepatitis B viral load with or without associated elevations in alanine aminotransferase and aspartate aminotransferase have been reported in patients with chronic hepatitis B virus (HBV) infections. Monitor and treat patients with chronic HBV infection according to clinical guidelines
- When discontinuing Jakafi, myeloproliferative neoplasm-related symptoms may return within one week. After discontinuation, some patients with myelofibrosis have experienced fever, respiratory distress, hypotension, DIC, or multi-organ failure. If any of these occur after discontinuation or while tapering Jakafi, evaluate and treat any intercurrent illness and consider restarting or increasing the dose of Jakafi. Instruct patients not to interrupt or discontinue Jakafi without consulting their physician. When discontinuing or interrupting Jakafi for reasons other than thrombocytopenia or neutropenia, consider gradual tapering rather than abrupt discontinuation
- Non-melanoma skin cancers (NMSC) including basal cell, squamous cell, and Merkel cell carcinoma have occurred. Perform periodic skin examinations
- Treatment with Jakafi has been associated with increases in total cholesterol, low-density lipoprotein cholesterol, and triglycerides. Assess lipid parameters 8-12 weeks after initiating Jakafi. Monitor and treat according to clinical guidelines for the management of hyperlipidemia
- Another JAK-inhibitor has increased the risk of major adverse cardiovascular events (MACE), including cardiovascular death, myocardial infarction, and stroke (compared to those treated with tumor TNF blockers) in patients with rheumatoid arthritis, a condition for which Jakafi is not indicated. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with Jakafi particularly in patients who are current or past smokers and patients with other cardiovascular risk factors. Patients should be informed about the symptoms of serious cardiovascular events and the steps to take if they occur
- Another JAK-inhibitor has increased the risk of thrombosis, including deep venous thrombosis (DVT), pulmonary embolism (PE), and arterial thrombosis (compared to those treated with TNF blockers) in patients with rheumatoid arthritis, a condition for which Jakafi is not indicated. In patients with myelofibrosis (MF) and polycythemia vera (PV) treated with Jakafi in clinical trials, the rates of thromboembolic events were similar in Jakafi and control treated patients. Patients with symptoms of thrombosis should be promptly evaluated and treated appropriately
- Another JAK-inhibitor has increased the risk of lymphoma and other malignancies excluding NMSC (compared to those treated with TNF blockers) in patients with rheumatoid arthritis, a condition for which Jakafi is not indicated. Patients who are current or past smokers are at additional increased risk. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with Jakafi, particularly in patients with a known secondary malignancy (other than a successfully treated NMSC), patients who develop a malignancy, and patients who are current or past smokers
- In myelofibrosis and polycythemia vera, the most common nonhematologic adverse reactions (incidence ≥15%) were bruising, dizziness, headache, and diarrhea. In acute graft-versus-host disease, the most common nonhematologic adverse reactions (incidence >50%) were infections (pathogen not specified) and edema. In chronic graft-versus-host disease, the most common nonhematologic adverse reactions (incidence >20%) were infections (pathogen not specified) and viral infections
- Avoid concomitant use with fluconazole doses greater than 200 mg. Dose modifications may be required when administering Jakafi with fluconazole doses of 200 mg or less, or with strong CYP3A4 inhibitors, or in patients with renal or hepatic impairment. Patients should be closely monitored and the dose titrated based on safety and efficacy
- Use of Jakafi during pregnancy is not recommended and should only be used if the potential benefit justifies the potential risk to the fetus. Women taking Jakafi should not breastfeed during treatment and for 2 weeks after the final dose
Please view Full Prescribing Information for Jakafi.
BAT=best available therapy; Hct=hematocrit; HU=hydroxyurea; JAK=janus kinase; PV=polycythemia vera; RESPONSE=Randomized study of Efficacy and Safety in POlycythemia vera with JAK iNhibitor ruxolitinib verSus bEst available care; WBC=white blood cell.
INDICATIONS AND USAGE
JAKAFI®/JAKAFI XR™ (ruxolitinib) is for treatment of intermediate or high-risk myelofibrosis (MF), including primary MF, post–polycythemia vera MF and post–essential thrombocythemia MF in adults.
JAKAFI/JAKAFI XR is for treatment of polycythemia vera (PV) in adults who have had an inadequate response to or are intolerant of hydroxyurea.
JAKAFI/JAKAFI XR is for treatment of steroid-refractory acute graft-versus-host disease (aGVHD) in adult and pediatric patients 12 years and older.
JAKAFI/JAKAFI XR is for treatment of chronic graft-versus-host disease (cGVHD) after failure of one or two lines of systemic therapy in adult and pediatric patients 12 years and older.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
Thrombocytopenia, Anemia and Neutropenia
- JAKAFI/JAKAFI XR can cause dose-related effects of thrombocytopenia, anemia and neutropenia. Perform a pre-treatment complete blood count (CBC) and monitor CBCs every 2 to 4 weeks until doses are stabilized, and then as clinically indicated.
- Manage thrombocytopenia by reducing the dose or temporarily interrupting JAKAFI/JAKAFI XR. Platelet transfusions may be necessary.
- Patients developing anemia may require blood transfusions and/or dose modifications of JAKAFI/JAKAFI XR.
- Severe neutropenia (ANC <0.5 × 109/L) was generally reversible by withholding JAKAFI/JAKAFI XR until recovery.
Risk of Infection
Tuberculosis
- Serious bacterial, mycobacterial, fungal and viral infections have occurred. Delay starting JAKAFI/JAKAFI XR until active serious infections have resolved. Observe patients receiving JAKAFI/JAKAFI XR for signs and symptoms of infection and manage promptly.
- Tuberculosis (TB) infection with JAKAFI/JAKAFI XR has been reported. Observe patients taking JAKAFI/JAKAFI XR for signs and symptoms of active TB and manage promptly. Prior to initiating, evaluate patients for TB risk factors and test those at higher risk for latent infection. Consult a physician with expertise in the treatment of TB before starting in patients with evidence of active or latent TB. Continuation during treatment of active TB should be based on the overall risk-benefit determination.
Progressive Multifocal Leukoencephalopathy
- Progressive multifocal leukoencephalopathy (PML) has occurred with JAKAFI/JAKAFI XR treatment. If PML is suspected, stop JAKAFI/JAKAFI XR and evaluate.
Herpes Zoster and Herpes Simplex
- Herpes zoster infection, reactivation and/or dissemination has been reported in patients receiving JAKAFI/JAKAFI XR. Advise patients about early signs and symptoms of herpes zoster and seek treatment. Monitor patients for the development of herpes simplex infections. If a patient develops evidence of dissemination of herpes simplex, consider interrupting treatment.
Hepatitis B
- Increases in hepatitis B viral load with or without associated elevations in alanine aminotransferase and aspartate aminotransferase have been reported in patients with chronic hepatitis B virus (HBV) infections.
Symptom Exacerbation Following Interruption or Discontinuation of Treatment
- When discontinuing JAK-Inhibitors, including JAKAFI/JAKAFI XR, myeloproliferative neoplasm-related signs and symptoms may flare. After discontinuation, some patients with myelofibrosis have experienced fever, respiratory distress, hypotension, disseminated intravascular coagulation (DIC), or multi-organ failure. If any of these occur after discontinuation or while tapering JAKAFI/JAKAFI XR, evaluate and treat any intercurrent illness and consider restarting or increasing the dose. Instruct patients not to interrupt or discontinue JAKAFI/JAKAFI XR without consulting their physician. When discontinuing or interrupting JAKAFI/JAKAFI XR for reasons other than life-threatening toxicities, consider gradual tapering rather than abrupt discontinuation.
Non-Melanoma Skin Cancer (NMSC)
- NMSC including basal cell, squamous cell, and Merkel cell carcinoma have occurred. Perform periodic skin examinations.
Lipid Elevations
- Treatment with JAKAFI/JAKAFI XR has been associated with increases in total cholesterol, low-density lipoprotein cholesterol, and triglycerides. Assess lipid parameters 8-12 weeks after initiation.
Major Adverse Cardiovascular Events (MACE)
- Another JAK-inhibitor has increased the risk of major adverse cardiovascular events (MACE), including cardiovascular death, myocardial infarction, and stroke (compared to those treated with TNF blockers), in patients with rheumatoid arthritis, a condition for which JAKAFI/JAKAFI XR is not indicated. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy, particularly in patients who are current or past smokers and patients with other cardiovascular risk factors. Patients should be informed about the symptoms of serious cardiovascular events and the steps to take if they occur.
Thrombosis
- Another JAK-inhibitor has increased the risk of thrombosis, including deep venous thrombosis (DVT), pulmonary embolism (PE), and arterial thrombosis (compared to those treated with TNF blockers), in patients with rheumatoid arthritis, a condition for which JAKAFI/JAKAFI XR is not indicated. In patients with myelofibrosis (MF) and polycythemia vera (PV), the rates of thromboembolic events were similar in JAKAFI/JAKAFI XR and control treated patients. Patients with symptoms of thrombosis should be promptly evaluated and treated appropriately.
Secondary Malignancies
- Another JAK-inhibitor has increased the risk of lymphoma and other malignancies, excluding NMSC (compared to those treated with TNF blockers), in patients with rheumatoid arthritis, a condition for which JAKAFI/JAKAFI XR is not indicated. Patients who are current or past smokers are at additional increased risk.
Adverse Reactions
- In myelofibrosis, the most common hematologic adverse reactions (incidence >20%) were thrombocytopenia and anemia. The most common nonhematologic adverse reactions (incidence ≥15%) were bruising, dizziness, headache and diarrhea.
- In polycythemia vera, the most common hematologic adverse reactions (incidence >20%) were thrombocytopenia and anemia. The most common nonhematologic adverse reactions (incidence ≥15%) were bruising, dizziness, headache and diarrhea.
- In acute graft-versus-host disease, the most common hematologic adverse reactions (incidence >50%) were anemia, thrombocytopenia and neutropenia. The most common nonhematologic adverse reactions (incidence >50%) were infections (pathogen not specified) and edema.
- In chronic graft-versus-host disease, the most common hematologic adverse reactions (incidence >35%) were anemia and thrombocytopenia. The most common nonhematologic adverse reactions (incidence ≥20%) were infections (pathogen not specified) and viral infections.
Drug Interactions
- Avoid concomitant use with fluconazole doses greater than 200 mg. Dose modifications may be required when administering fluconazole doses of 200 mg or less, or with strong CYP3A4 inhibitors, or in patients with renal or hepatic impairment. Patients should be closely monitored and the dose titrated based on safety and efficacy.
Pregnancy
- Use during pregnancy is not recommended and should only be used if the potential benefit justifies the potential risk to the fetus. Women taking JAKAFI/JAKAFI XR should not breastfeed during treatment and for 2 weeks after the final dose.
Please see Full Prescribing Information for JAKAFI and JAKAFI XR.
INDICATIONS AND USAGE
JAKAFI®/JAKAFI XR™ (ruxolitinib) is for treatment of intermediate or high-risk myelofibrosis (MF), including primary MF, post–polycythemia vera MF and post–essential thrombocythemia MF in adults.
JAKAFI/JAKAFI XR is for treatment of polycythemia vera (PV) in adults who have had an inadequate response to or are intolerant of hydroxyurea.
JAKAFI/JAKAFI XR is for treatment of steroid-refractory acute graft-versus-host disease (aGVHD) in adult and pediatric patients 12 years and older.
JAKAFI/JAKAFI XR is for treatment of chronic graft-versus-host disease (cGVHD) after failure of one or two lines of systemic therapy in adult and pediatric patients 12 years and older.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
Thrombocytopenia, Anemia and Neutropenia
- JAKAFI/JAKAFI XR can cause dose-related effects of thrombocytopenia, anemia and neutropenia. Perform a pre-treatment complete blood count (CBC) and monitor CBCs every 2 to 4 weeks until doses are stabilized, and then as clinically indicated.
- Manage thrombocytopenia by reducing the dose or temporarily interrupting JAKAFI/JAKAFI XR. Platelet transfusions may be necessary.
- Patients developing anemia may require blood transfusions and/or dose modifications of JAKAFI/JAKAFI XR.
- Severe neutropenia (ANC <0.5 × 109/L) was generally reversible by withholding JAKAFI/JAKAFI XR until recovery.
Risk of Infection
Tuberculosis
- Serious bacterial, mycobacterial, fungal and viral infections have occurred. Delay starting JAKAFI/JAKAFI XR until active serious infections have resolved. Observe patients receiving JAKAFI/JAKAFI XR for signs and symptoms of infection and manage promptly.
- Tuberculosis (TB) infection with JAKAFI/JAKAFI XR has been reported. Observe patients taking JAKAFI/JAKAFI XR for signs and symptoms of active TB and manage promptly. Prior to initiating, evaluate patients for TB risk factors and test those at higher risk for latent infection. Consult a physician with expertise in the treatment of TB before starting in patients with evidence of active or latent TB. Continuation during treatment of active TB should be based on the overall risk-benefit determination.
Progressive Multifocal Leukoencephalopathy
- Progressive multifocal leukoencephalopathy (PML) has occurred with JAKAFI/JAKAFI XR treatment. If PML is suspected, stop JAKAFI/JAKAFI XR and evaluate.
Herpes Zoster and Herpes Simplex
- Herpes zoster infection, reactivation and/or dissemination has been reported in patients receiving JAKAFI/JAKAFI XR. Advise patients about early signs and symptoms of herpes zoster and seek treatment. Monitor patients for the development of herpes simplex infections. If a patient develops evidence of dissemination of herpes simplex, consider interrupting treatment.
Hepatitis B
- Increases in hepatitis B viral load with or without associated elevations in alanine aminotransferase and aspartate aminotransferase have been reported in patients with chronic hepatitis B virus (HBV) infections.
Symptom Exacerbation Following Interruption or Discontinuation of Treatment
- When discontinuing JAK-Inhibitors, including JAKAFI/JAKAFI XR, myeloproliferative neoplasm-related signs and symptoms may flare. After discontinuation, some patients with myelofibrosis have experienced fever, respiratory distress, hypotension, disseminated intravascular coagulation (DIC), or multi-organ failure. If any of these occur after discontinuation or while tapering JAKAFI/JAKAFI XR, evaluate and treat any intercurrent illness and consider restarting or increasing the dose. Instruct patients not to interrupt or discontinue JAKAFI/JAKAFI XR without consulting their physician. When discontinuing or interrupting JAKAFI/JAKAFI XR for reasons other than life-threatening toxicities, consider gradual tapering rather than abrupt discontinuation.
Non-Melanoma Skin Cancer (NMSC)
- NMSC including basal cell, squamous cell, and Merkel cell carcinoma have occurred. Perform periodic skin examinations.
Lipid Elevations
- Treatment with JAKAFI/JAKAFI XR has been associated with increases in total cholesterol, low-density lipoprotein cholesterol, and triglycerides. Assess lipid parameters 8-12 weeks after initiation.
Major Adverse Cardiovascular Events (MACE)
- Another JAK-inhibitor has increased the risk of major adverse cardiovascular events (MACE), including cardiovascular death, myocardial infarction, and stroke (compared to those treated with TNF blockers), in patients with rheumatoid arthritis, a condition for which JAKAFI/JAKAFI XR is not indicated. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy, particularly in patients who are current or past smokers and patients with other cardiovascular risk factors. Patients should be informed about the symptoms of serious cardiovascular events and the steps to take if they occur.
Thrombosis
- Another JAK-inhibitor has increased the risk of thrombosis, including deep venous thrombosis (DVT), pulmonary embolism (PE), and arterial thrombosis (compared to those treated with TNF blockers), in patients with rheumatoid arthritis, a condition for which JAKAFI/JAKAFI XR is not indicated. In patients with myelofibrosis (MF) and polycythemia vera (PV), the rates of thromboembolic events were similar in JAKAFI/JAKAFI XR and control treated patients. Patients with symptoms of thrombosis should be promptly evaluated and treated appropriately.
Secondary Malignancies
- Another JAK-inhibitor has increased the risk of lymphoma and other malignancies, excluding NMSC (compared to those treated with TNF blockers), in patients with rheumatoid arthritis, a condition for which JAKAFI/JAKAFI XR is not indicated. Patients who are current or past smokers are at additional increased risk.
Adverse Reactions
- In myelofibrosis, the most common hematologic adverse reactions (incidence >20%) were thrombocytopenia and anemia. The most common nonhematologic adverse reactions (incidence ≥15%) were bruising, dizziness, headache and diarrhea.
- In polycythemia vera, the most common hematologic adverse reactions (incidence >20%) were thrombocytopenia and anemia. The most common nonhematologic adverse reactions (incidence ≥15%) were bruising, dizziness, headache and diarrhea.
- In acute graft-versus-host disease, the most common hematologic adverse reactions (incidence >50%) were anemia, thrombocytopenia and neutropenia. The most common nonhematologic adverse reactions (incidence >50%) were infections (pathogen not specified) and edema.
- In chronic graft-versus-host disease, the most common hematologic adverse reactions (incidence >35%) were anemia and thrombocytopenia. The most common nonhematologic adverse reactions (incidence ≥20%) were infections (pathogen not specified) and viral infections.
Drug Interactions
- Avoid concomitant use with fluconazole doses greater than 200 mg. Dose modifications may be required when administering fluconazole doses of 200 mg or less, or with strong CYP3A4 inhibitors, or in patients with renal or hepatic impairment. Patients should be closely monitored and the dose titrated based on safety and efficacy.
Pregnancy
- Use during pregnancy is not recommended and should only be used if the potential benefit justifies the potential risk to the fetus. Women taking JAKAFI/JAKAFI XR should not breastfeed during treatment and for 2 weeks after the final dose.
Please see Full Prescribing Information for JAKAFI and JAKAFI XR.