Jakafi® (ruxolitinib) targets the primary driver of PV1
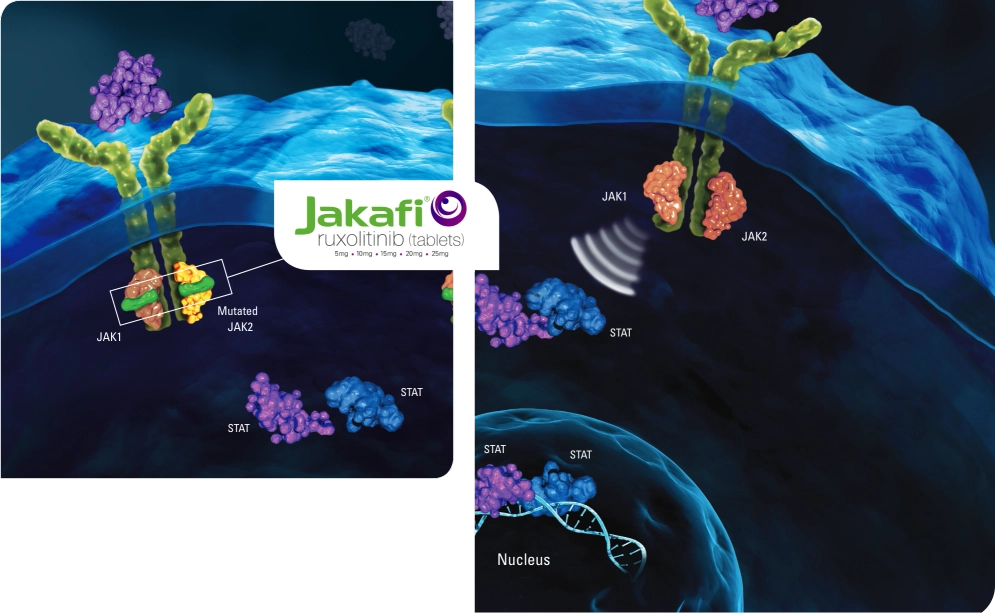
JAK1
of key proinflammatory cytokines2
JAK2
hematopoietic growth factors2
Hct=hematocrit; JAK=Janus kinase; PV=polycythemia vera; STAT=signal transducer and activator of transcription; WBC=white blood cell.
References: 1. Jakafi [package insert]. Wilmington, DE: Incyte Corporation. 2. Quintás-Cardama A, Vaddi K, Liu P, et al. Preclinical characterization of the selective JAK1/2 inhibitor INCB018424: therapeutic implications for the treatment of myeloproliferative neoplasms. Blood. 2010;115(15):3109-3117.

